- Home
- What is Acid Rain
- Acid Rain Creation
- About Sulfur Dioxide
- About Nitrogen Oxide
- Environmental Effects
- Problem Areas
- What's Being Done
- What You Can Do at Home
- Our Action Plan
- Survey Results
- Summing it Up!
- Sources
- Contact Us
What is Acid Rain?
Acid precipitation is a form of acid deposition. Acids are dissolved in rain, fog, dew, or snow, where the precipitation has a PH of less then 5.6, which is the PH of normal rain water.
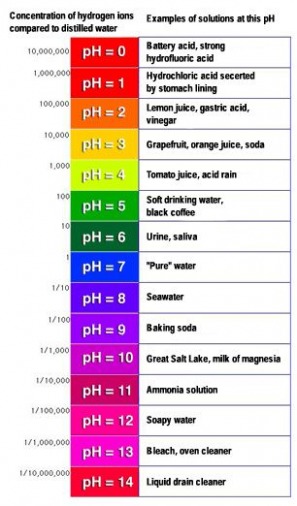
The PH scale is used to measure the acidity of liquids. If you are not familiar with the PH scale, below is an example.
0 = maximum acidity
7 = neutral point in the middle of the scale
14 = maximum alkalinity (the opposite of acidity)